Red Mud is produced during the Bayer Process. With this process, we can extract the aluminum (oxy)hydroxides from bauxites and get alumina, which eventually can be smelted and give aluminum.
Bayer Process
In 1888, Karl Josef Bayer developed and patented a process, which has become the cornerstone of the aluminium production industry worldwide. The Bayer process, as it has become known, is used for refining bauxite to smelting grade alumina (aluminum oxide), the precursor to aluminium. Typically, depending upon the quality of the ore, between 1.9 and 3.6 tonnes of bauxite is required to produce 1 tonne of alumina.
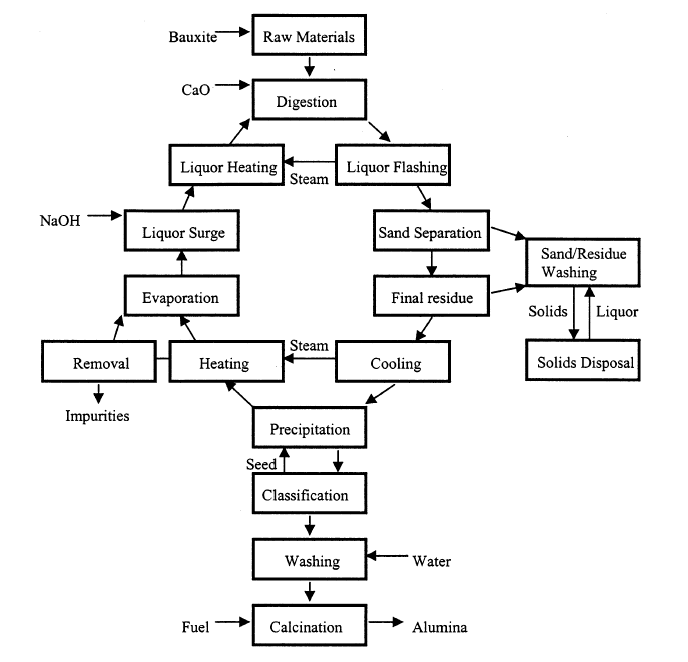
The Bayer process is a cyclic one and is often called Bayer cycle. It involves four steps: digestion, clarification, precipitation, and calcination.
[Source: A. R. Hind, S. K. Bhargava, Stephen C. Grocott, “The surface chemistry of Bayer process solids: a review”, Colloids and Surfaces A : Physicochem. Eng. Aspects, 146 (1999) 359–374. Other schematic diagrams of Bayer process (flowsheets) are available in the works published by B.I. Whittington, 1996 and Y. L. Sidrak, 2001].
In the following animated picture you have another approach of the Bayer process
[Source: http://www.specialty-aluminas.alcan.com/gardanne/webgardannev2.nsf/vwUrl/MondeAlumine_Bayer_VI]
Digestion
In the first step, bauxite is ground, slurried with a solution of caustic soda (sodium hydroxide), and pumped into large pressure tanks called digesters, where the ore is subjected to steam heat and pressure. The specific conditions depend on the quality of bauxite used (i.e. the type of aluminum (oxy)hydoxides present. The sodium hydroxide reacts with the aluminous minerals of bauxite to form a saturated solution of sodium aluminate; insoluble impurities, called red mud (RM), remain in suspension and are separated in the clarification step. The following reactions describe this step:
Clarification
Following digestion, the mixture is passed through a series of pressure-reducing tanks (called blow-off tanks), where the solution is flashed to atmospheric pressure. The next step in the process is to separate the insoluble red mud from the sodium aluminate solution. Coarse material (e.g., beach sand) is removed in crude cyclones called sand traps. Finer residue is settled in raking thickeners with the addition of synthetic flocculants, and solids in the thickener overflow are removed by cloth filters. These residues are then washed, combined, and discarded. The clarified solution is further cooled in heat exchangers, enhancing the degree of supersaturation of the dissolved alumina, and pumped into tall, silolike precipitators.
Precipitation
Sizable amounts of aluminum hydroxide crystals are added to the solution in the precipitators as seeding to hasten crystal separation. The seed crystals attract other crystals and form agglomerates; these are classified into larger product-sized material and finer material that is recycled as seed. The product-sized agglomerates of aluminum hydroxide crystals are filtered and washed to remove entrained caustic or solution. The following reaction takes place:
Calcination
The agglomerates of aluminum hydroxide are calcined in rotary kilns or stationary fluidized-bed flash calciners at temperatures in excess of 960°C (1,750°F). Free water and water that is chemically combined are driven off, leaving commercially pure alumina. The following reaction describes this step:
As can be seen in the flowsheet, lime (CaO) is also introduced in the Bayer process. According to a review on this issue by B.I. Whittington 1996, it can enhance the extraction of alumina (improving the dissolution of boehmite and diaspore or the conversion of aluminogoethite), control liquor impurities (desilication, causticisation or phosphorous control), assist with the removal of impurities from the pregnant liquor (liquor “polishing”) and minimise soda losses in the red mud (formation of alternate desilication products or calcium titanates).
Alumina produced by the Bayer process is quite pure, containing only a few hundredths of 1 percent of iron and silicon. The major impurity, residual soda, is present at levels of 0.2 to 0.6 percent. In addition to being the primary raw material for producing metallic aluminum, alumina itself is an important chemical. It is used widely in the chemical, refractories, ceramic, and petroleum industries.
Refining four tons of bauxite yields about two tons of alumina. A typical alumina plant, using the Bayer process, can produce 4,000 tons of alumina per day. The cost of alumina can vary widely, depending on the plant size and efficiency, on labour costs and overhead, and on the cost of bauxite.
Bibliography
Encyclopedia Britannica
A. R. Hind, S. K. Bhargava, Stephen C. Grocott, “The surface chemistry of Bayer process solids: a review”, Colloids and Surfaces A : Physicochem. Eng. Aspects, 146 (1999) pp. 359–374
http://www.specialty-aluminas.alcan.com/gardanne/webgardannev2.nsf/vwUrl/MondeAlumine_Bayer_VI
B.I. Whittington, “The chemistry of CaO and Ca(OH)2 relating to the Bayer process”, Hydrometallurgy 43 (1996), pp. 13-35
Yousry L. Sidrak, “Dynamic Simulation and Control of the Bayer Process. A Review”, Ind. Eng. Chem. Res. 40, (2001), pp. 1146-56
Text written by Y. Pontikes. Reflects only personal opinions. Last updated July 13, 2005